Medtronic (NY:MDT)
101.28
+0.20
(+0.19%)
Streaming Delayed Price
Updated: 10:17 AM EST, Feb 12, 2026
Add to My Watchlist
Press Releases about Medtronic







Via AB Newswire
Topics
Economy

There’s a New Way to Treat AFib - Here’s What You Should Know
September 28, 2025
Via NewsUSA
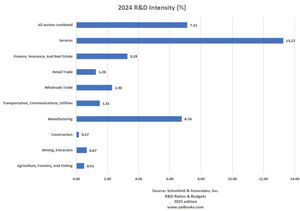
2024 R&D Spending and Intensity Benchmarks
September 08, 2025
Via PRLog
Topics
Supply Chain













Via Get News
Topics
Supply Chain






Stock Quote API & Stock News API supplied by www.cloudquote.io
Quotes delayed at least 20 minutes.
By accessing this page, you agree to the Privacy Policy and Terms Of Service.
Quotes delayed at least 20 minutes.
By accessing this page, you agree to the Privacy Policy and Terms Of Service.
© 2025 FinancialContent. All rights reserved.


