FDA withdraws Covid antibody treatment Evusheld because it's not effective against 93% of subvariants
January 26, 2023 at 23:20 PM EST
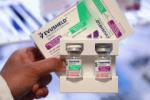
Many take Evusheld as an additional layer of protection because the vaccines do not trigger a strong immune response for them.
